What is Catalytic Oxidation of Carbon Monoxide?
Catalytic oxidation of carbon monoxide is a highly efficient purification technology that utilizes catalysts to lower reaction activation energy, enabling the targeted conversion of carbon monoxide and oxygen into non-toxic carbon dioxide under mild conditions. The core of this technology lies in the selection of appropriate catalysts and their adaptation to specific operating conditions. Mainstream catalysts are broadly categorized into two groups—precious metals and non-precious metals—each suited to the distinct requirements of different application scenarios. This technology offers numerous advantages, including low ignition temperatures, thorough purification, and safety and stability; it is widely applied in fields such as industrial waste gas treatment and air purification in enclosed spaces, serving as a pivotal solution for addressing carbon monoxide pollution.

I. What is Catalytic Oxidation of Carbon Monoxide? (Definition and Analysis)
Catalytic oxidation of carbon monoxide is a gas purification technology based on catalytic reactions. Its core principle involves utilizing a catalyst to facilitate a redox reaction between toxic carbon monoxide (CO) and oxygen (O₂)—without the need for high temperatures or open flames—thereby achieving the targeted conversion of CO into non-toxic and harmless carbon dioxide (CO₂) and effectively accomplishing the removal and purification of carbon monoxide. Compared to traditional CO treatment technologies—such as physical adsorption and high-temperature combustion—this technology offers significant advantages, including low energy consumption, high conversion efficiency, and the absence of secondary pollution. Consequently, it is currently the preferred technology for the deep purification of CO in both industrial and residential sectors; it can consistently reduce outlet CO concentrations to below 5 ppm, achieving purification efficiencies ranging from 95% to over 99%.
II. Core Principles of Catalytic Oxidation of Carbon Monoxide
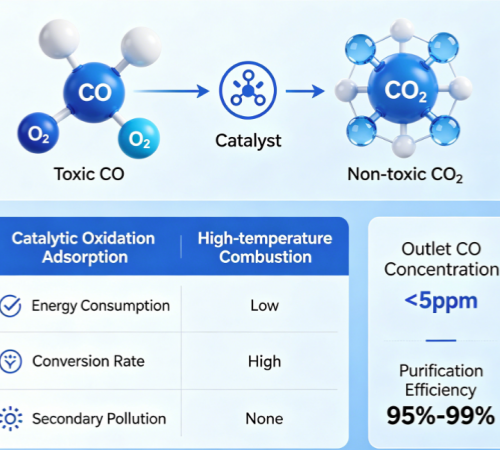
The essence of catalytic oxidation of carbon monoxide lies in the catalytic acceleration of the CO oxidation reaction. Its core mechanism involves the catalyst lowering the reaction's activation energy—thereby breaking down the energy barrier between CO and O₂—and facilitating the rapid occurrence of the reaction under mild conditions. The chemical equation for this reaction is: 2CO + O₂ (Catalyst) → 2CO₂. The entire process involves no open flames and poses no risk of explosion; moreover, the heat generated during the reaction can be recovered and utilized, further reducing energy consumption. In terms of reaction mechanism, CO molecules first adsorb onto the active sites of the catalyst; once activated, they react with oxygen species adsorbed on the catalyst surface to form CO₂. The newly formed CO₂ then desorbs from the catalyst surface, completing the catalytic cycle. Crucially, the catalyst itself is not consumed during the reaction, serving solely to accelerate the process.
III. Mainstream Catalyst Types for Carbon Monoxide Catalytic Oxidation

Catalysts constitute the core of carbon monoxide catalytic oxidation technology; their performance directly determines the reaction efficiency, operating temperature, and applicable scenarios. Currently, mainstream catalysts fall into two primary categories, each with distinct characteristics tailored to different operating conditions. Precious metal catalysts utilize active components such as platinum (Pt), palladium (Pd), and gold (Au), which are supported on carriers such as alumina (Al₂O₃) or ceria (CeO₂). These catalysts offer significant advantages, including high activity at low temperatures, strong resistance to sulfur and moisture, and excellent stability. Consequently, they are well-suited for complex operating environments—such as those found in the chemical industry and waste incineration facilities—though they are associated with higher costs. Non-precious metal catalysts, centered on materials such as copper-manganese oxides and cobalt oxides, are characterized by their low cost and abundant availability. They are broadly categorized into ambient-temperature types (e.g., Hopcalite catalysts) and medium-to-high-temperature types. These are respectively suited for ambient-temperature environments—such as mines and rescue capsules—and for medium-to-high-temperature industrial settings, such as steel sintering operations; however, their low-temperature activity and resistance to catalyst poisoning still require further optimization.
IV. Typical Application Scenarios for Catalytic Oxidation of Carbon Monoxide
Leveraging its characteristics of high efficiency, safety, and energy conservation, catalytic oxidation technology for carbon monoxide has been widely adopted across numerous fields to address CO pollution issues in diverse settings. In the industrial sector, it is primarily utilized for the deep purification of CO found in steel sintering flue gas, blast furnace gas, and chemical process tail gas. This technology can be integrated with desulfurization and denitrification systems to achieve comprehensive control of multiple pollutants, while simultaneously recovering reaction heat to reduce production energy consumption. In enclosed spaces—such as mine rescue capsules, underground parking garages, and submarines—it enables the rapid removal of CO, thereby ensuring personnel safety. In the civil and environmental sectors, it is applied in areas such as the purification of exhaust from gas water heaters, fire-fighting self-rescuers, and Regenerative Catalytic Oxidation (RCO) systems, facilitating the efficient removal of CO and ensuring compliance with environmental and safety standards.
author:kaka
date:2026/4/22





 minstrong
minstrong